^ Xia, Kang Zheng, Xianxu Yuan, Jianan Liu, Cong Gao, Hao Wu, Qiang Sun, Jian ()."Alkaline-earth metal (Mg) polynitrides at high pressure as possible high-energy materials". ^ Wei, Shuli Li, Da Liu, Zhao Li, Xin Tian, Fubo Duan, Defang Liu, Bingbing Cui, Tian (2017)."Emergence of Novel Polynitrogen Molecule-like Species, Covalent Chains, and Layers in Magnesium–Nitrogen Mg x N y Phases under High Pressure". Zhang, Litong Frapper, Gilles (June 2017). ^ Yu, Shuyin Huang, Bowen Zeng, Qingfeng Oganov, Artem R.^ "Summary of Classification and Labelling".Magnesium nitride has also been applied to synthesize boron nitride nanocrystals and nitrides of aluminum and Group 3 It has also been proposed as an intermediate in a fossil-fuel-free nitrogen fixation process. These lumps could scratch a polished block of boron carbide, something only diamond was known to do.įrom the smell of ammonia, caused by the reaction of magnesium nitride with the moisture in the air, he deduced that the magnesium metal had reacted with the boron nitride to form magnesium nitride, which was the true catalyst. When he examined the wire under a microscope, he found tiny dark lumps clinging to it. 
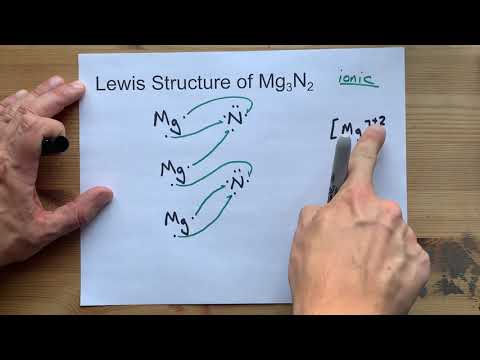
Out of desperation and curiosity (he called it the "make the maximum number of mistakes" approach ), he added some magnesium wire to the hexagonal boron nitride and gave it the same pressure and heat treatment. He had already tried all the logical catalysts (for instance, those that catalyze the synthesis of diamond), but with no success. was trying to convert the hexagonal form of boron nitride into the cubic form by a combination of heat, pressure, and a catalyst. Magnesium nitride was the catalyst in the first practical synthesis of borazon (cubic boron nitride). When isolating argon, William Ramsay passed dry air over copper to remove oxygen and over magnesium to remove the nitrogen, forming magnesium nitride. This Mg 2N 4 compound was recovered to ambient conditions, along with the N 4 4− units, marking only the fourth polynitrogen entity bulk stabilized at ambient conditions. The Mg 2N 4 is composed of exotic cis-tetranitrogen N 4 4− species with N-N bond orders close to one. These include the Mg 2N 4 and MgN 4 solids which both become thermodynamically stable near 50 GPa. Thermal decomposition of magnesium nitride gives magnesium and nitrogen gas (at 700-1500 ☌).Īt high pressures, the stability and formation of new nitrogen-rich nitrides (N/Mg ratio equal or greater to one) were suggested and later discovered. In fact, when magnesium is burned in air, some magnesium nitride is formed in addition to the principal product, magnesium oxide. Magnesium nitride reacts with water to produce magnesium hydroxide and ammonia gas, as do many metal nitrides. By passing dry nitrogen over heated magnesium:ģ Mg + N 2 → 800 ∘ C Mg 3 N 2 Chemistry.At room temperature and pressure it is a greenish yellow powder. Magnesium nitride, which possesses the chemical formula Mg 3N 2, is an inorganic compound of magnesium and nitrogen. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
